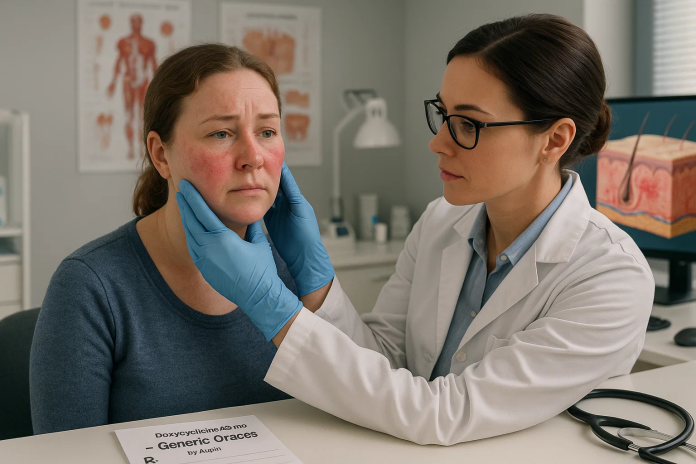
Indian pharmaceutical powerhouse Lupin Ltd. has achieved a major milestone by launching the first generic version of Oracea (doxycycline capsules, 40 mg) in the United States, a move that strengthens both its dermatology portfolio and its foothold in the lucrative U.S. generics market.
The Lupin Generic Oracea is now approved by the U.S. Food and Drug Administration (FDA) and is therapeutically equivalent to Oracea by Galderma Laboratories, a branded drug widely used for treating inflammatory lesions of rosacea. This chronic skin condition affects over 16 million Americans and is characterized by redness, swelling, and acne-like bumps, especially on the face.
Why This Launch Matters
According to IQVIA™ (March 2024), Oracea recorded U.S. sales of approximately $145 million — representing a major opportunity for cost-effective, high-quality generic competition. As the first generic entrant, Lupin is positioned to capture a significant portion of this market before others follow suit.
Unlike traditional antibiotics, Oracea delivers low-dose doxycycline at a sub-antimicrobial concentration to reduce inflammation without contributing to antibiotic resistance. Lupin’s generic offers patients the same benefit at a fraction of the cost, supporting broader access to safe and effective treatment.

Lupin’s Strategic Global Expansion
The launch of Lupin Generic Oracea aligns with the company’s long-term strategy of expanding its complex generics portfolio, especially in specialty and dermatology segments — areas with higher margins and growing global demand.
“This product is a reflection of our commitment to offering advanced treatments at affordable costs,” said Vinita Gupta, CEO of Lupin, in a recent press release. “It strengthens our position in the dermatology market and demonstrates our capability to bring first-to-market generics that improve patient access.”
Implications for U.S. Patients and Providers
The U.S. healthcare system has been battling high drug prices, particularly in chronic conditions like rosacea where branded therapies dominate. Lupin’s FDA-approved alternative provides physicians and pharmacists a cost-efficient substitute while maintaining therapeutic efficacy. This shift could also encourage insurers to widen reimbursement options for rosacea treatment.
What’s Next for Lupin?
With this successful launch, Lupin is expected to accelerate submissions of complex generics, particularly in respiratory, ophthalmology, and dermatology sectors. The company is also exploring biosimilars and digital therapeutics to diversify its product line further.